Pipeline
In addition to the services that iCeutica provides to pharmaceutical partners, iCeutica also utilizes the SoluMatrix™ Fine Particle Technology to develop new products that it actively develops itself and with collaborators.
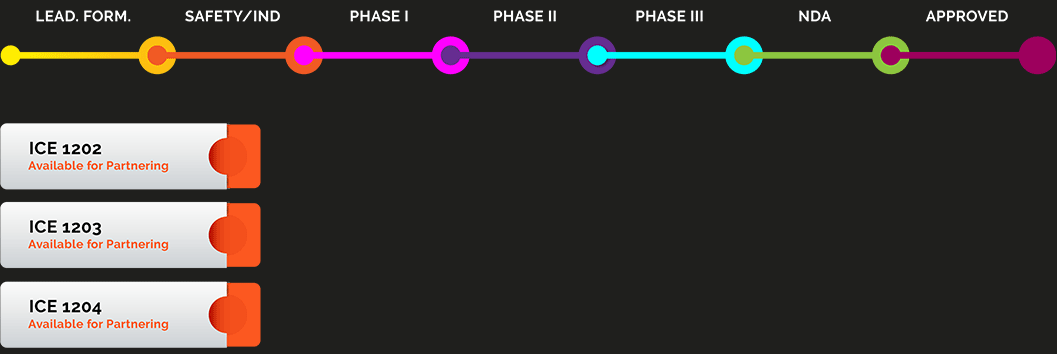
iCeutica is advancing a pipeline of product candidates that address broad indications, including pain and inflammation, oncology, and respiratory diseases. Our products are poised for rapid success, due to quick and clear regulatory and commercial pathways, and are in areas of significant commercial opportunity and unmet need.
iCeutica has successfully worked in partnership with a number of companies including Iroko Pharmaceuticals (with NSAIDs) and Churchill Pharmaceuticals (with oncology agents) to develop and gain approval for a number of FDA approved medicines.